Thermal extraction with carrier gas
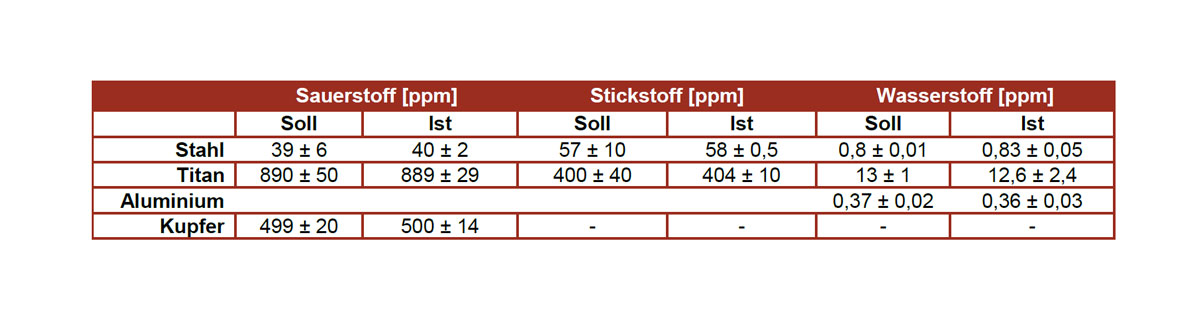
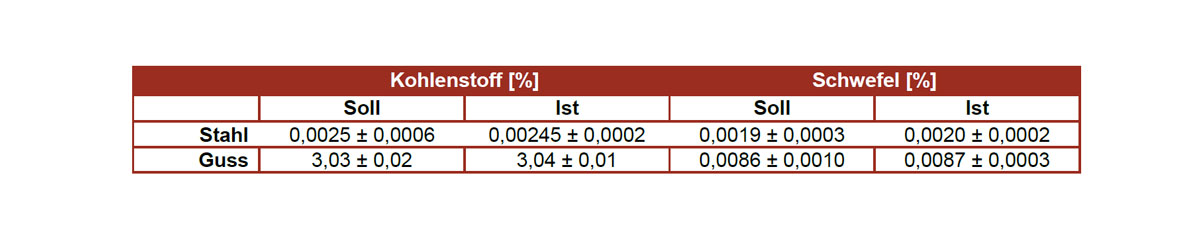
Analysis of the elements nitrogen (N), oxygen (O), hydrogen (H), carbon (C) and sulfur (S)
Methods: Thermal extraction with carrier gas (TGHE) on the Elementrac ONHp analyzer Combustion analysis (C/S) on the CS-200 analyzer
| Element | Nachweisgrenzen* |
|---|---|
| Sauerstoff | 4 ppm – 20 ppm |
| Stickstoff | 4 ppm – 35 ppm |
| Wasserstoff | 0,1 ppm – 4 ppm |
| Kohlenstoff | 13 ppm – 20 ppm |
| Schwefel | 4 ppm |
*Matrix- und Probenabhängig |
The sample preparation and the analyzes are usually carried out according to standards such as e.g. B. ASTM E1409, ASTM E1447, DIN EN ISO 15350, ASTM E1019, ASTM E1941, ASTM E2792, DIN EN 2003-10 and / or DIN EN ISO 956. Of course, TAZ GmbH is accredited for these standards.
Operating principle ONH
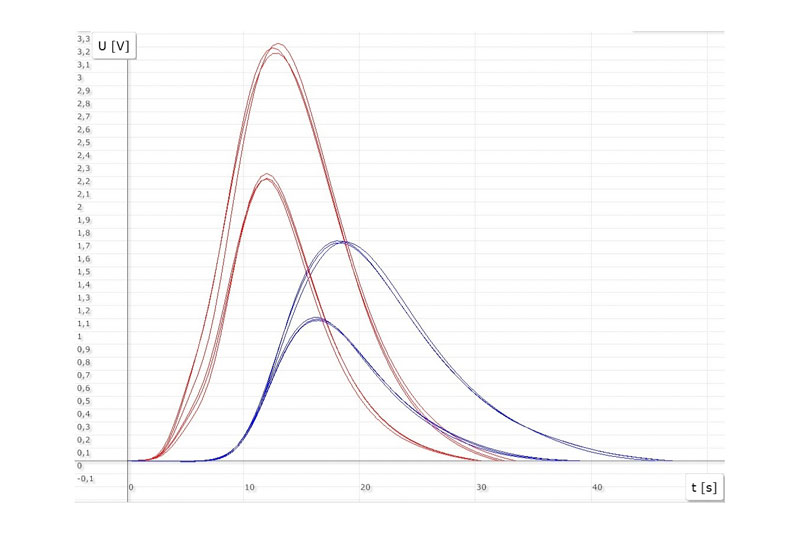
For the analysis of the elements O, N and H, the metal sample is thermally decomposed. The metal sample is heated in a graphite crucible in an induction furnace. Temperatures of up to 3000 °C can be reached here. Helium or nitrogen is used as carrier gas. The released gases, whereby the oxygen O reacts with the carbon C of the graphite crucible to form carbon monoxide CO, flow through various stations (catalysis, absorber, etc.) of the measuring system with the help of the carrier gas flow and are detected in the corresponding measuring cells. The oxygen content is determined via the CO2 content in an infrared measuring cell. The molecular hydrogen and nitrogen is measured with a thermal conductivity cell. Since the thermal conductivity cell is used to determine hydrogen and nitrogen, the analyzer works in two different operating modes and can therefore detect either nitrogen or hydrogen.
- ON mode: Determination of oxygen and nitrogen with the carrier gas helium
- OH mode: Determination of oxygen and hydrogen with the carrier gas nitrogen
Functional principle CS
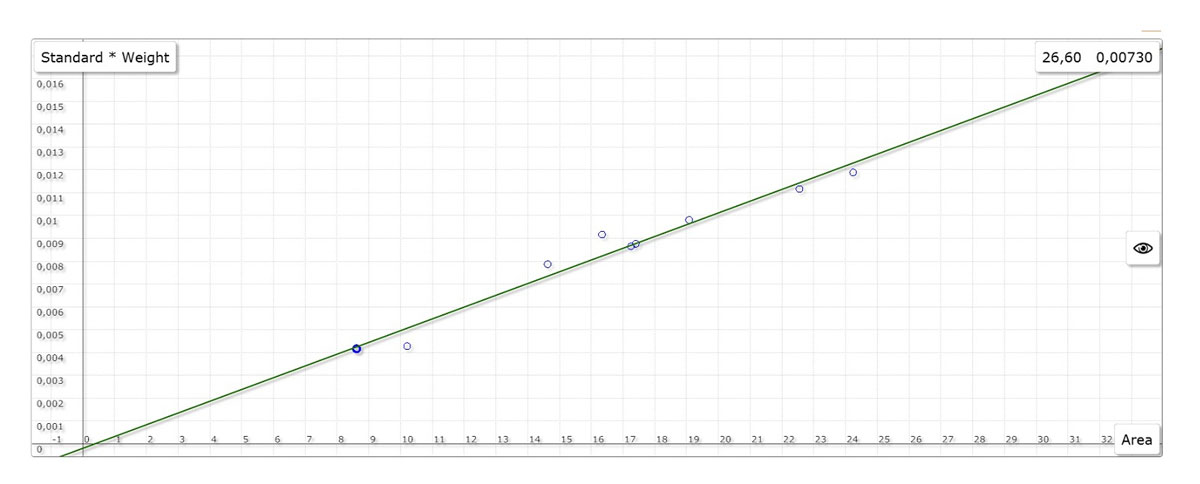
A high-frequency combustion of the metal samples takes place at approx. 2300 °C in a pure oxygen atmosphere. During combustion, the released carbon components are oxidized to CO/CO₂ and sulfur components to SO₂. Dust and water are then filtered out of the combustion gas. The combustion gases are passed on to the corresponding infrared measuring cells in the carrier gas flow (oxygen). Compact samples as well as chips, wires or powder can be analyzed for both ONH and CS.